China’s First Self-Developed ECMO Medical Equipment Cleared for Release
Since the pandemic broke out in early 2020, extracorporeal membrane oxygenation (ECMO), a initially-help procedure made use of to supply clients prolonged respiratory and cardiac help, has captivated significant awareness for its higher price tag tag. On January 4, China’s very first domestic ECMO devices was approved for community sale.
Following examination, China’s Health-related Solutions Administration urgently authorized the software of Shenzhen Chinabridge Healthcare Technological know-how Co., Ltd. for extracorporeal cardiopulmonary support auxiliary devices and disposable membrane oxygenator kits. These products and solutions have unbiased intellectual residence legal rights, and their general performance generally reaches the global amount of related merchandise.
The core factors of ECMO are membrane lungs and blood pumps, which participate in the part of replacing lungs and hearts, respectively. ECMO is regarded for being life-saving, miraculous and extremely pricey. The rate of an ECMO device commonly ranges from 1 million yuan to 3 million yuan ($145,878 – $437,636). According to a procurement announcement on June 23, 2022, Huashan Healthcare facility put in 2.5 million yuan on a person set of ECMO. In addition to the procurement expense, the equipment demands expensive auxiliary gadgets through procedure.
In accordance to Chinabridge Healthcare, the common price of employing a established of ECMO equipment for patients is 50,000 yuan, and the every day cure price is about 10,000-20,000 yuan, which will past one or two weeks, resulting in the overall cost to be as large as 200,000 yuan.
The approval of Chinabridge Medical’s ECMO items signifies a important stage in China’s initiatives to conclude its whole dependence on imports of the technologies. In simple fact, Chinabridge Healthcare is a organization that has only existed for a lot more than 4 a long time. It was launched in May 2018 by medical engineering authorities studying in Germany. In July 2022, it accomplished A-spherical financing of about 100 million yuan, with Shenzhen Capital Group as the major trader.
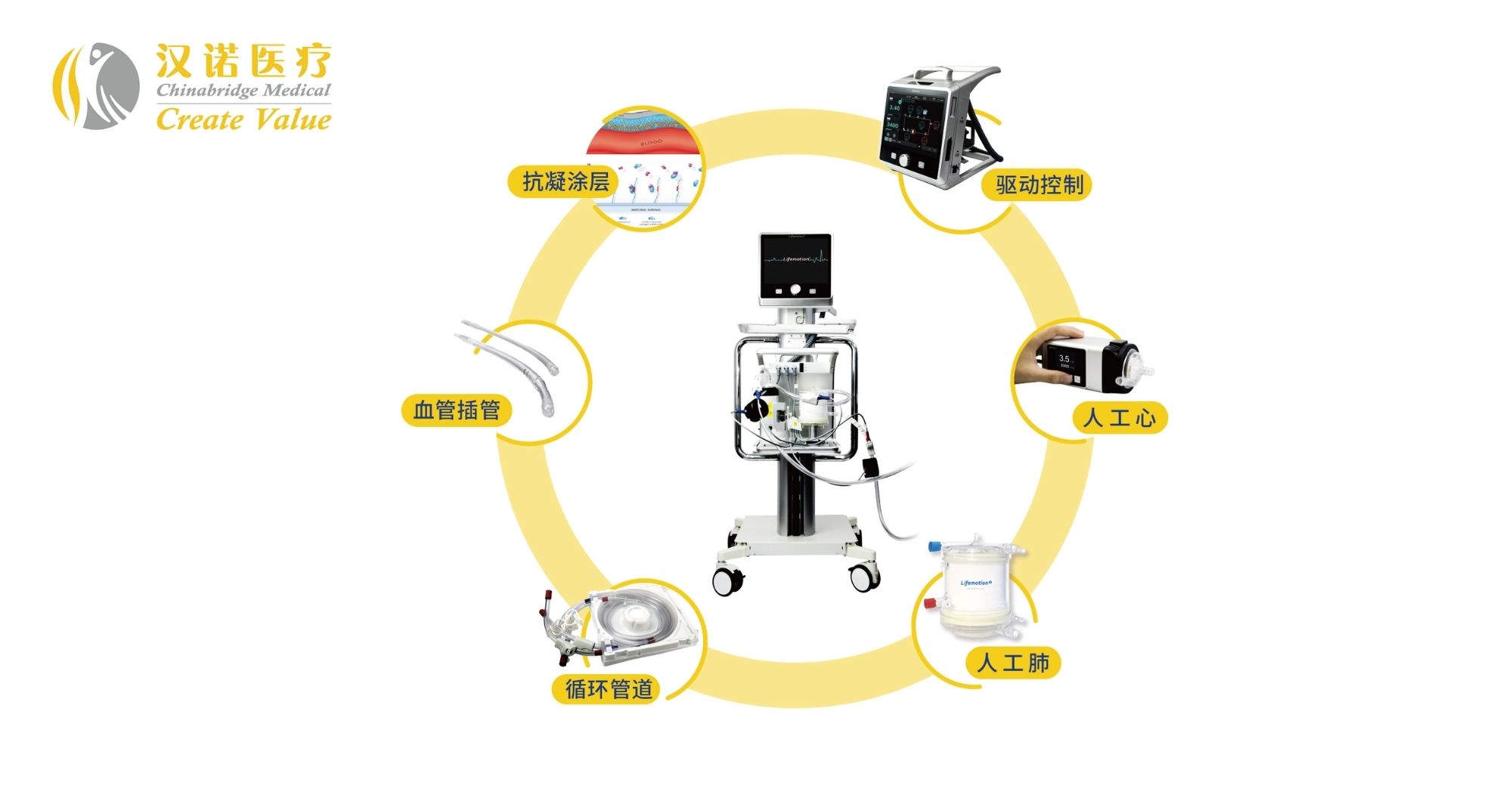
The progress of Chinabridge Professional medical is inextricably connected both equally with Germany and with several main technologies in the ECMO discipline. In April 2020, Chinabridge Health-related cooperated with an innovative R&D workforce of innovative lifestyle assistance from Germany to develop a lifetime very first help method referred to as Lifemotion.
According to information unveiled by Chinabridge Healthcare in November 2019, Liu Yang, the latest chairman of Chinabridge Clinical, is also director of the China Challenge Division of the Germany-China Medication Association. Close cooperation with the German scientific investigate crew might be the crucial rationale why Chinabridge Healthcare has been equipped to capture up in the ECMO field.
SEE ALSO: Chinese Citizens Flock to Health and fitness Applications in Bid to Acquire COVID-19 Meds
Considering that 2022, Chinabridge Medical’s ECMO system has carried out more than 10 scientific trials in several hospitals in Beijing, Sichuan, Guangdong and Jiangsu. The 2nd-era ECMO program of Chinabridge Health-related is also underneath progress. The enterprise has 53 patent applications, which include more than 20 utility patents and a lot more than 20 creation patents. 2022 is the 12 months in which the corporation utilized for the most patents.
